Estimated Read Time: 9–11 minutes
Summary: This article explores the emerging research behind low dose naltrexone and dry eyes, particularly in patients living with diabetes. It explains how diabetes-related dry eye may involve deeper biological disruptions affecting tear production, corneal sensitivity, and inflammatory signaling. It reviews preclinical studies, early human trial data, and the proposed role of the OGF–OGFr pathway in ocular surface balance. It also discusses how compounded topical low dose naltrexone may be considered under prescriber guidance when conventional supportive options provide incomplete relief. Finally, this article outlines the importance of sterile ophthalmic compounding and collaborative care for patients and clinicians exploring individualized dry eye strategies.
Exploring Options for Diabetes-Related Dry Eye
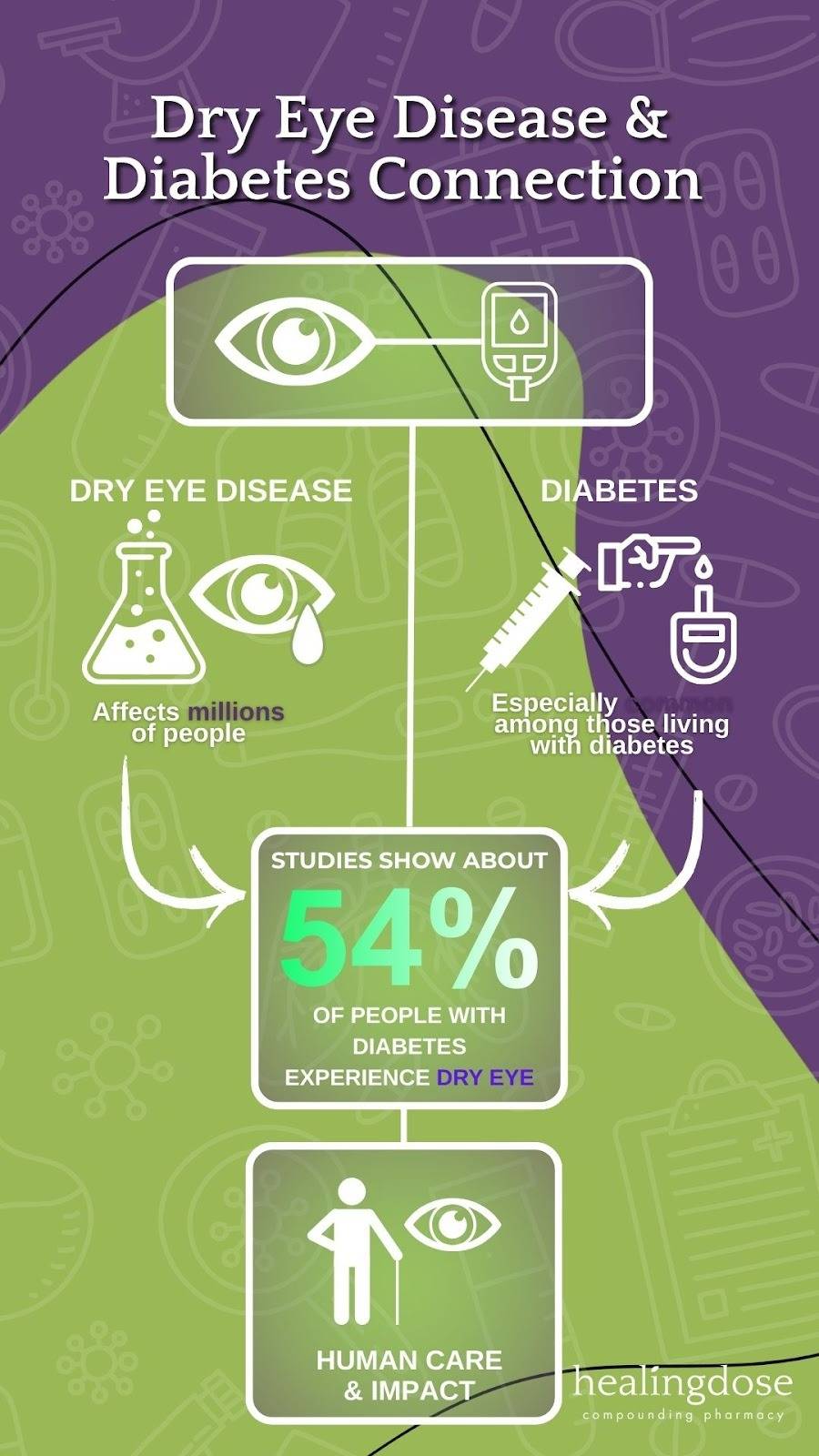
Diabetic dry eye is an increasingly recognized complication of diabetes mellitus, affecting an estimated 54% of Americans living with the disease. As diabetes prevalence continues to rise across the United States — including Michigan, where more than 1 in 10 adults are diagnosed with diabetes and many more remain undiagnosed — ocular surface complications are becoming more common in both primary eye care and specialty settings.
Chronic hyperglycemia (high blood sugar) can disrupt lacrimal gland function, impair corneal nerve integrity, and alter inflammatory signaling pathways, contributing to reduced tear production, unstable tear film, and decreased corneal sensitivity. For many patients with diabetes, dry eye symptoms are not simply mild irritation but a persistent source of visual fluctuation, discomfort, and diminished quality of life, often proving more complex and refractory than non-diabetic dry eye disease.
As awareness of the complex neurobiological and regulatory components of diabetic dry eye increases, more eye care practitioners have begun exploring compounded approaches that extend beyond traditional tear replacement and surface anti-inflammatory therapies. Among these, Low Dose Naltrexone (LDN) has generated clinical interest due to its potential effects on the opioid growth factor (OGF)–OGFr pathway.
In an eye drop form, research suggests topical naltrexone may influence pathways involved in Dry Eye Disease.
How the OGF–OGFr Pathway Affects Tear Production
The opioid growth factor (OGF)–opioid growth factor receptor (OGFr) axis is a regulatory system involved in controlling cell growth, tissue repair, and surface integrity — including within the cornea and lacrimal functional unit.
What Is OGF?
OGF (also known as [Met5]-enkephalin) is an endogenous opioid peptide naturally produced in the body. Unlike opioid peptides involved in pain signaling, OGF primarily functions as a regulator of cell proliferation.
It helps maintain balance by slowing excessive cell division and coordinating normal epithelial turnover.
What Does OGFr Do?
OGFr is the specific receptor that binds OGF. When OGF binds to OGFr, it:
- Enters the cell nucleus
- Influences gene expression
- Helps regulate the cell cycle
- Maintains controlled corneal epithelial renewal
In healthy ocular tissue, this interaction maintains corneal surface stability and supports normal epithelial repair.
What Happens in Diabetes?
In diabetes, research suggests that the OGF–OGFr pathway becomes dysregulated.
Studies in diabetic models have shown:
- Elevated OGF levels
- Increased OGFr expression
- Excessive inhibitory signaling
This overactivity may:
- Suppress normal corneal epithelial cell replication
- Slow surface healing
- Reduce tear production
- Alter corneal nerve sensitivity
The result is impaired ocular surface homeostasis — contributing to tear film instability and reduced corneal integrity.
In other words, the regulatory “brake” becomes too strong.
Where Does Low Dose Naltrexone Fit In?
Low Dose Naltrexone (LDN) is an opioid receptor antagonist. When applied topically:
- It transiently blocks OGFr
- This interruption is temporary (short duration blockade)
- The body responds with a compensatory upregulation of OGF and OGFr production
- This may help normalize regulatory signaling over time
What’s Happening?
→ LDN transiently blocks OGFr
→ compensatory upregulation of OGF and OGFr
→ restoration of homeostatic cell turnover regulation.
Rather than permanently suppressing the pathway, LDN appears to “reset” or rebalance the system through short-term receptor blockade followed by endogenous feedback adjustments.
In diabetic animal models, this has been associated with:
- Improved tear production
- Increased corneal epithelial cell proliferation
- Improved corneal sensitivity
- Reduced inflammatory markers
Source: BMC Ophthalmology
Why This May Matter for Tear Production
The lacrimal gland, corneal nerves, and epithelial cells function as an integrated unit (the lacrimal functional unit). If epithelial turnover and nerve signaling are impaired:
- Tear secretion may decline
- Reflex tearing can be reduced
- Surface repair slows
- Symptoms persist despite lubrication
From research:
Dry eye disease involves disruption of the lacrimal functional unit — an integrated system consisting of the lacrimal glands, ocular surface, corneal nerves, and associated immune pathways. Diabetes may impair multiple components of this unit, including neural signaling and epithelial maintenance, contributing to tear film instability and altered corneal sensation.
Diabetes is also associated with corneal neuropathy, characterized by reduced corneal sensitivity and impaired epithelial healing. Experimental studies have demonstrated that topical naltrexone administration restored corneal sensation toward non-diabetic levels in both type 1 and type 2 diabetic models.
Prescription Low Dose Naltrexone Eye Drops
As interest in opioid receptor modulation at the ocular surface has grown, attention has also turned to how low dose naltrexone can be delivered appropriately for ophthalmic use. Because there is no commercially available FDA-approved naltrexone eye drop, any topical ophthalmic preparation must be compounded pursuant to a valid prescription.
Compounding allows the medication to be formulated at low concentrations suitable for ocular application, prepared in sterile conditions, and tailored to the specific dosing and tolerability considerations determined by the prescribing clinician. In this setting, sterile ophthalmic compounding becomes essential — not only to achieve the desired concentration, but to ensure proper formulation, stability, and patient safety for use on the ocular surface.
Understanding Compounded Low Dose Naltrexone (LDN) Eye Drops
- No Commercially Available Product
- There is currently no FDA-approved, commercially manufactured naltrexone ophthalmic solution for dry eye disease.
- Any topical ophthalmic LDN preparation must therefore be compounded pursuant to a valid prescription.
- Need for Low, Ocular-Appropriate Concentrations
- Oral naltrexone products are manufactured at significantly higher systemic doses.
- Ophthalmic use requires low, precisely measured concentrations suitable for the sensitive ocular surface.
- Sterile Preparation Is Essential
- Ophthalmic medications must be compounded in sterile environments to reduce the risk of contamination.
- Preparation must follow USP <797> sterile compounding standards.
- Customized Formulation Considerations
- Concentration and dosing frequency are determined by the prescribing clinician.
- Preservative-free options may be considered when clinically appropriate.
- Formulation choices may be adjusted based on patient tolerability.
- Clinical Oversight Required
- Compounded LDN eye drops are dispensed only under prescriber guidance.
- Ongoing monitoring is recommended to assess response and tolerability.
What is Research Saying About Topical Naltrexone & Diabetic Dry Eye
Naltrexone Interacting With Diabetic Dry Eye Pathways
Published in Investigative Ophthalmology & Visual Science, researchers wanted to better understand why dry eye develops in the context of type 1 diabetes. They explored whether blocking specific regulatory pathways at the ocular surface could influence those changes.
Researcher Findings
- Diabetes was linked to measurable changes in tear production and corneal sensitivity, reflecting disruption at the ocular surface.
- The OGF–OGFr regulatory system appeared altered in the diabetic state, suggesting a shift in pathways involved in corneal cell turnover.
- After topical naltrexone was introduced, tear production and corneal sensitivity moved toward levels observed in non-diabetic controls.
- Markers related to corneal epithelial cell proliferation increased, indicating potential normalization of surface renewal processes.
- Inflammatory markers within corneal tissue were reduced, alongside shifts in specific nerve-related markers linked to corneal sensation.
Evaluating Naltrexone Eye Drop
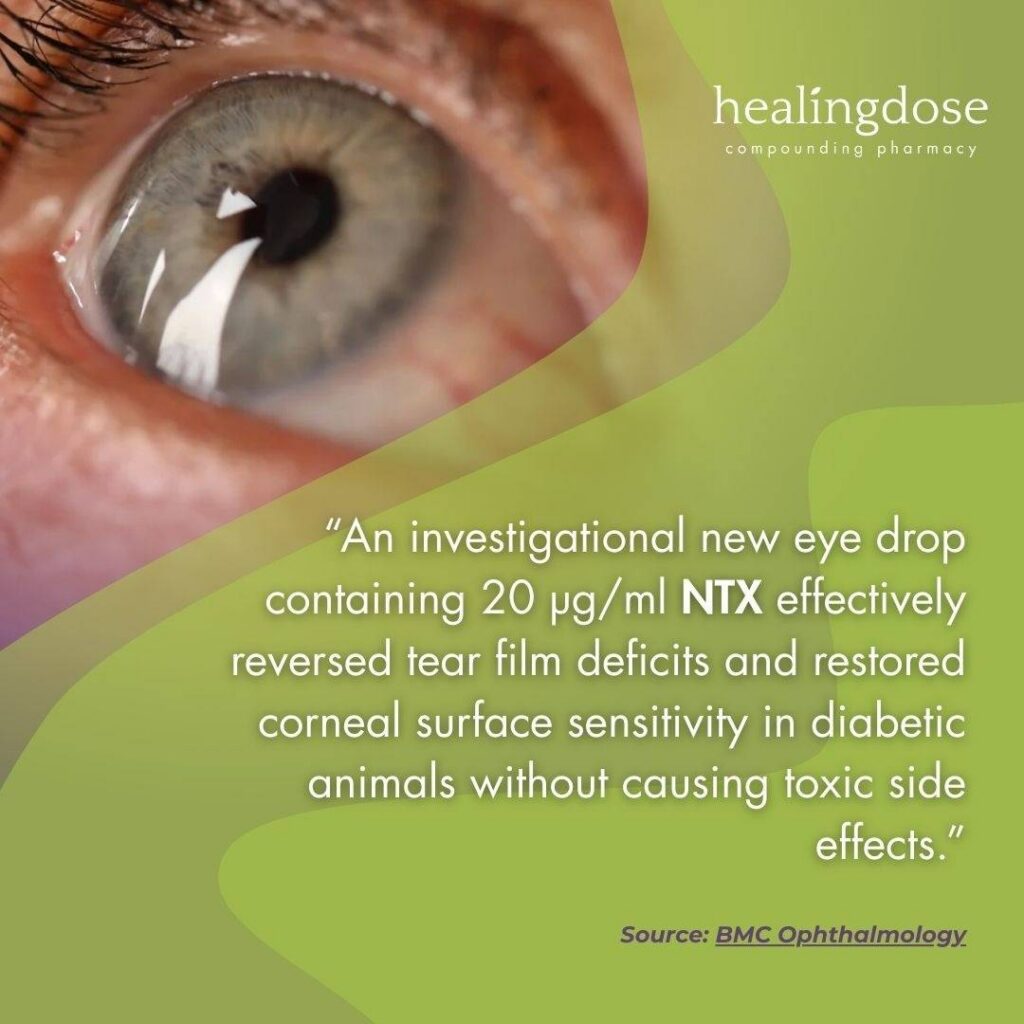
Investigators explored whether a specific ophthalmic formulation containing naltrexone could influence key features of dry eye disease in a diabetic animal model. Published in BMC Ophthalmology, this investigation focused on a 20-µg/mL naltrexone formulation and examined its potential effects on tear production and corneal sensitivity and its safety in rats and rabbits.
Investigator Findings
- In diabetic rats, the eyes of subjects showed increases in tear volume within hours after a single application compared with controls.
- Over the course of 10 days of twice-daily dosing, corneal surface sensitivity also moved closer to levels seen in non-diabetic animals.
- In safety evaluations with naïve rats and rabbits receiving the drug twice daily for 30 days, no visible ocular pathology, irritation, redness, or overt signs of discomfort were observed.
- Measures such as intraocular pressure remained within normal ranges after exposure, suggesting the formulation was well-tolerated in animal eyes.
Phase 2 Human Clinical Trial of Topical Naltrexone for Dry Eye in Diabetic Subjects
A Phase 2, double-masked, placebo-controlled clinical trial was conducted to explore the safety, tolerability, and exploratory signals of a low-dose (0.002%) topical naltrexone ophthalmic solution. Signs and symptoms of dry eye were observed over a 29-day period among adult participants with diabetes.
Reported Observations at Day 29
- The total visual analog scale pain score trended lower in the naltrexone group compared with placebo, suggesting a possible directional reduction in perceived ocular discomfort.
- Average tear osmolarity values were lower in the naltrexone arm, moving closer to ranges generally considered less suggestive of dry eye.
- Measurements showed a small directional shift toward higher corneal sensitivity in the naltrexone group, indicating relative preservation of sensation.
These early human observations are exploratory in nature and may inform clinician-directed discussions regarding off-label compounded ophthalmic options in select cases, rather than establishing definitive therapeutic conclusions.
Clinical Scenarios Where Compounded Topical LDN May Be Considered in Diabetic Dry Eye
Compounded topical low dose naltrexone may be considered under prescriber guidance in cases such as:
- Persistent Dry Eye Symptoms Despite Standard Therapy
- Ongoing dryness, irritation, or fluctuating vision despite consistent use of artificial tears, lubricants, or prescribed anti-inflammatory agents.
- Symptoms Disproportionate to Surface Findings
- Significant ocular discomfort, burning, or pain with minimal staining, redness, or visible surface damage on examination.
- Features Suggestive of Neuropathic Ocular Pain
- Altered corneal sensitivity, heightened pain response, or sensory abnormalities commonly seen in diabetes-related nerve dysfunction.
- Recurrent or Fluctuating Symptoms in Type 1 Diabetes
- Cyclical or unpredictable symptom patterns despite stable glycemic control and routine ocular management.
- Objective Indicators of Ocular Surface Dysregulation
- Abnormal tear osmolarity, tear-film instability, or measurable corneal sensitivity changes suggesting broader regulatory disruption.
- Incomplete Response to Conventional Anti-Inflammatory Approaches
- Partial improvement with corticosteroids, cyclosporine, lifitegrast, or other therapies, but continued symptom burden.
- Sensitivity or Intolerance to Certain Ophthalmic Ingredients
- Stinging or irritation from preserved eye drops prompting consideration of alternative compounded formulations.
- Stinging or irritation from preserved eye drops prompting consideration of alternative compounded formulations.
This content is intended solely for educational purposes and does not constitute medical advice, diagnosis, or treatment recommendations. Compounded ophthalmic preparations are dispensed pursuant to a valid prescription and individualized clinical determination, and outcomes may vary based on patient-specific factors.
For Prescribers: Exploring Low Dose Naltrexone
Low dose naltrexone (LDN) is a versatile compounded medication that can be prepared in multiple dosage forms based on clinical need, including capsules, oral liquids, sublingual drops, topical creams, and sterile ophthalmic solutions. Beyond investigational ophthalmic applications, LDN has been prescribed off-label in various settings for conditions involving chronic pain, autoimmune disorders, inflammatory syndromes, and certain neurologic concerns. As a compounded medication, the formulation, strength, and route of administration are determined by the prescribing clinician to align with individual patient factors and therapeutic goals.
Compounded medications are prepared by a licensed pharmacist based on a valid prescription and are not reviewed or approved by the FDA.
Experienced Ophthalmic Compounding Pharmacy in Michigan
ALSO SERVING FL, IN, MN, OH, & WI
At Healing Dose Compounding Pharmacy, our pharmacists work closely with patients and prescribers throughout Michigan and beyond to support individualized care in complex clinical situations. Through sterile ophthalmic compounding and custom formulations, we serve as a collaborative partner when commercially available options may not fully align with a patient’s needs.
Topical naltrexone would not replace established dry eye therapies such as artificial tears, punctal occlusion, anti-inflammatory agents, or immunomodulators, but may be explored in select cases under specialist guidance.

Why Michigan Patients and Prescribers Choose HDRx
- PCAB- and ACHC-accredited, with sterile compounding performed in accordance with USP <797> standards
Ophthalmic compounded formulations at HDRx are prepared in ISO-classified cleanroom environments in accordance with USP <797>, with:
- Sterility testing protocols
- Endotoxin monitoring where applicable
- Appropriate beyond-use dating
- Preservative-free options when clinically indicated
- Sterility testing protocols
- Experience in crafting custom ophthalmic formulations, including ingredient-specific combinations selected with tolerability and clinical intent in mind
- Dedicated Provider Portal to support efficient collaboration and communication
- Prescription order forms to simplify the compounding process
- Licensed to ship prescription compounds to Michigan, Florida, Indiana, Minnesota, Ohio, and Wisconsin, with over-the-counter products available nationwide
If you’re a patient living with diabetes and struggling with ongoing dry eye, consult with your eye care provider about compounded ophthalmic options and how they may fit your situation.
If you’re a prescriber, HDRx collaborates closely with clinicians to support formulation considerations, sterile compounding, and ongoing care coordination. Our team is available to assist with compounded ophthalmic preparations under appropriate clinical guidance.
Contact our team to learn more about compounded medications, ophthalmic services, and provider resources.
References
- Diaz, D., Sassani, J. W., Zagon, I. S., & McLaughlin, P. J. (2025). Reversal of diabetic dry eye by topical opioid receptor blockade follows dual pathways. Investigative Ophthalmology & Visual Science, 66(3), 24. Link
- Dry Eye with Diabetes | American Diabetes Association. (n.d.-b). Link
- McLaughlin, P. J., Sassani, J. W., Titunick, M. B., & Zagon, I. S. (2019). Efficacy and safety of a novel naltrexone treatment for dry eye in type 1 diabetes. BMC Ophthalmology, 19(1), 35. Link
- McLaughlin, P. J., Sassani, J. W., & Zagon, I. S. (2020). Naltrexone as a novel therapeutic for diabetic corneal complications. Journal of cellular immunology, 2(2), 42. Link
- Stern, M. E., Gao, J., Siemasko, K. F., Beuerman, R. W., & Pflugfelder, S. C. (2004). The role of the lacrimal functional unit in the pathophysiology of dry eye. Experimental eye research, 78(3), 409-416. Link
- Zagon, I. S., Klocek, M. S., Sassani, J. W., & McLaughlin, P. J. (2009). Topical naltrexone reverses dry eye and restores corneal sensation in diabetes mellitus. Archives of ophthalmology, 127(11), 1468. Link
- Zagon, I. S., Sassani, J. W., Immonen, J. A., & McLaughlin, P. J. (2014). Ocular surface abnormalities related to type 2 diabetes are reversed by the opioid antagonist naltrexone. Clinical & experimental ophthalmology, 42(2), 159-168. Link